Clinical Trials
In our clinical research trials we are studying novel treatments for diseases associated with the aging of the immune system such as cancer and frailty. Here are some of the active trials we have going on.
1. Aging of the immune system/Immune Senescence
PROTOCOL 001.16-BMSCTI: “Phase 1/2 Clinical Study Evaluating the Safety and Efficacy of Fresh Frozen Plasma (GMFFP) from young healthy donors ages 18 to 35, who have received Granulocyte – Colony Stimulating Factor (G-CSF) to Ameliorate Frailty and Enhance the Immune Risk Profile in Older Individuals”
The primary purpose of this study is to determine the safety and tolerability of 12, once monthly transfusions of Granulocyte – Colony Stimulating Factor (G-CSF) Mobilized Fresh Frozen Plasma (GMFFP) harvested from young healthy donors and given to older, frail individuals who are at risk due to unhealthy aging and who will then have a subsequent 12-month follow-up period. The secondary objective is to determine the efficacy in older, frail individuals of 12, once monthly transfusions of GMFFP (Granulocyte – Colony Stimulating Factor (G-CSF) Mobilized Fresh Frozen Plasma harvested from young, healthy donors), and a subsequent 12-month follow-up period, to improve the Immune Risk Profile, (“IRP”), cognitive function (MME), quality of life (OPQOL-35), Frailty Index, (“FI”), associated with unhealthy aging in the treated subjects.
This study has an IND [Investigational New Drug Application] with the FDA [United States Food and Drug Administration]. The study is IRB [Institutional Review Board] approved.
2. Solid Tumors/Cancer
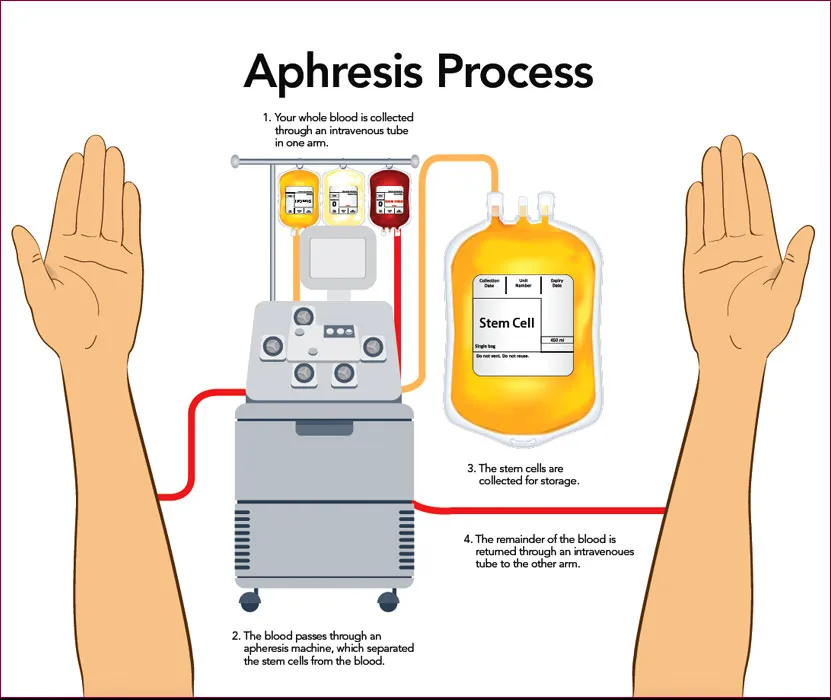
A Phase I/II Study For Use of White Blood Cells From Healthy Donor Participants to Treat Solid Tumors
The primary purpose of this study is to determine the safety of White Cell Transfer (WCT), i.e. intravenous infusion given from HLA-mismatched healthy donors. The secondary objective of this study is to determine the efficacy of White Cell Transfer in terms of disease response, i.e. complete response, partial response, stable disease, and progressive disease.
This investigational new cancer therapy was developed from the laboratory research done by Dr. Zheng Cui MD., Ph.D. who identified that a particular strain of lab mouse (SR/CR mice) was cancer resistant. Dr. Cui’s research concluded that the cancer resistance was mediated by a specific population of leukocytes (granulocytes and monocytes) and that these cells, when transfused to a mouse with cancer, would result in complete remission of the cancer.
The aim of this clinical trial is to replicate in humans the same results observed in mice and, ultimately, lead to an effective, nontoxic treatment for newly diagnosed patients as well as patients who can no longer benefit from conventional treatments.
This study has an IND [Investigational New Drug Application] with the FDA [United States Food and Drug Administration]. The study is IRB [Institutional Review Board] approved.
Request An Appointment
Please fill out the form to request an appointment. We will contact you shortly to confirm the exact day/time.